United States (EN)
Select your region or country.
ORCA-Flash4.0 V3 Digital CMOS camera
C13440-20CU
We’ve advanced our camera technology, so you can advance your science
Building on our extensive experience with high performance scientific cameras and advanced imaging applications, Hamamatsu introduces the new ORCA®-Flash4.0 V3. This one camera expertly handles applications ranging from the acquisition of beautiful scientific images to experiments that demand detection, quantification and speed. With on-board FPGA processing enabling intelligent data reduction, highly refined in-camera, pixel-level calibrations, increased USB3.0 frame rates, purposeful and innovative triggering capabilities, patented lightsheet read out modes and individual camera noise characterization the ORCA-Flash4.0 V3 is the precision instrument for imaging.
ORCA is a registered trademark of Hamamatsu Photonics K.K. (China, EU, France, Germany, Japan, UK, USA).
Features
- Calibrated for Quantitative Accuracy
- Flexibility for Customized Data Control
- Patented Tools for Advanced Imaging: Lightsheet Readout Mode
- Focus on the Relevant Data
- Powerful Triggering for Synchronizationa
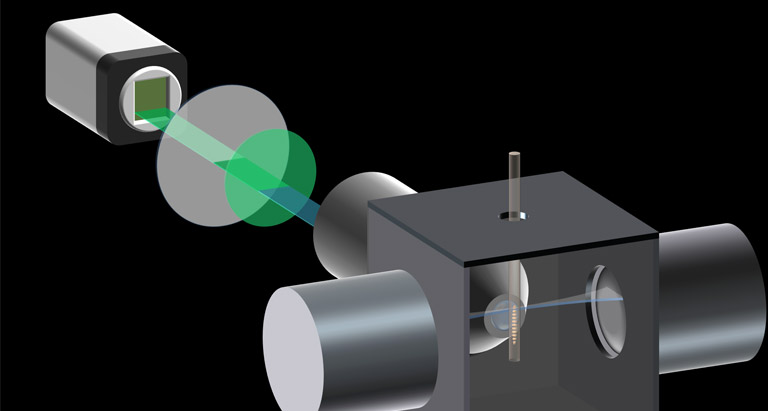
Lightsheet Readout Mode [Patented]
“Lightsheet Readout Mode” is a unique and patented feature of Hamamatsu sCMOS cameras which can improve signal to noise ratios in Lightsheet microscopy.
For more information about the principle and features of Lightsheet Readout Mode, please see details from below.
Movie gallery
Publications
ORCA-Flash4.0
| Authors | Title | Source |
|---|---|---|
| Xuanze Chen, Zhiping Zeng, Rongqin Li, Boxin Xue, Peng Xi, Yujie Sun | Superior performance with sCMOS over EMCCD in super-resolution optical fluctuation imaging | Journal of Biomedical Optics |
| Luchang Li, Mengting Li, Zhaoning Zhang, Zhen-Li Huang | Assessing low-light cameras with photon transfer curve method | Journal of Innovative Optical Health Sciences |
| S Hayashi, Y Okada | Ultrafast superresolution fluorescence imaging with spinning disk confocal microscope optics | Molecular Biology of the Cell |
| Matthias Mehling, Tino Frank, Cem Albayrak, Sava? Tay | Real-time tracking, retrieval and gene expression analysis of migrating human T cells | Lab on a Chip |
| Ying S. Hu, Maxwell Zimmerley, Yu Li, Robin Watters, and Hu Cang | Single-Molecule Super-Resolution Light-Sheet Microscopy | ChemPhysChem |
| Peter W Winter and Hari Shroff | Faster fluorescence microscopy: advances in high speed biological imaging | Current Opinion in Chemical Biology |
| Ronny Forster, Hui-Wen Lu-Walther, Aurelie Jost, Martin Kielhorn, Kai Wicker, and Rainer Heintzmann | Simple structured illumination microscope setup with high acquisition speed by using a spatial light modulator | Optics express |
| Jeffrey P. Nguyen, Frederick B. Shipley, Ashley N. Linder, George S. Plummer, Joshua W. Shaevitz, and Andrew M. Leifer | Whole-brain calcium imaging with cellular resolution in freely behaving C. elegans | Neurons and Cognition |
| Zong W, Zhao J, Chen X, Lin Y, Ren H, Zhang Y, Fan M, Zhou Z, Cheng H, Sun Y, Chen L. | Large-field high-resolution two-photon digital scanned light-sheet microscopy | Cell Res. 2014 Sep 26. doi: 10.1038/cr.2014.124. [Epub ahead of print] |
| Vladimirov N, Mu Y, Kawashima T, Bennett DV, Yang CT, Looger LL, Keller PJ, Freeman J, Ahrens MB. | Light-sheet functional imaging in fictively behaving zebrafish | Nat Methods. 2014 Jul 27. doi: 10.1038/nmeth.3040. [Epub ahead of print] |
| Mickoleit M, Schmid B, Weber M, Fahrbach FO, Hombach S, Reischauer S, Huisken J. | High-resolution reconstruction of the beating zebrafish heart | Nat Methods. 2014 Jul 20. doi: 10.1038/nmeth.3037. [Epub ahead of print] |
| Vettenburg T, Dalgarno HI, Nylk J, Coll-Lladó C, Ferrier DE, Čižmár T, Gunn-Moore FJ, Dholakia K. | Light-sheet microscopy using an Airy beam | Nat Methods. 2014 May;11(5):541-4. doi: 10.1038/nmeth.2922. Epub 2014 Apr 6. |
| Juette MF, Terry DS1, Wasserman MR, Zhou Z, Altman RB, Zheng Q, Blanchard SC. | The bright future of single-molecule fluorescence imaging | Curr Opin Chem Biol. 2014 Jun 20;20C:103-111. doi: 10.1016/j.cbpa.2014.05.010. |
| Menon M, Askinazi OL, Schafer DA. | Dynamin2 organises lamellipodial actin networks to orchestrate lamellar actomyosin | PLoS One. 2014 Apr 7;9(4):e94330. doi: 10.1371/journal.pone.0094330. eCollection 2014. |
| Gualda EJ, Vale T, Almada P, Feijó JA, Martins GG, Moreno N. | OpenSpinMicroscopy: an open-source integrated microscopy platform | Nat Methods. 2013 Jul;10(7):599-600. doi: 10.1038/nmeth.2508. Epub 2013 Jun 9. |
| Fahrbach FO, Voigt FF, Schmid B, Helmchen F, Huisken J. | Rapid 3D light-sheet microscopy with a tunable lens | Opt Express. 2013 Sep 9;21(18):21010-26. doi: 10.1364/OE.21.021010. |
| Fullerton S, Bennett K, Toda E, Takahashi T. | Optimization of precision localization microscopy using CMOS camera technology | Proc. SPIE 8228, Single Molecule Spectroscopy and Superresolution Imaging V, 82280T (February 9, 2012); doi:10.1117/12.906336; http://dx.doi.org/10.1117/12.906336 |
| Fullerton S, Bennett K, Toda E, Takahashi T. | Camera simulation engine enables efficient system optimization for super-resolution imaging | Proc. SPIE 8228, Single Molecule Spectroscopy and Superresolution Imaging V, 822811 (February 9, 2012); doi:10.1117/12.906346; http://dx.doi.org/10.1117/12.906346 |
| Singh AP, Krieger JW, Buchholz J, Charbon E, Langowski J, Wohland T. | The performance of 2D array detectors for light sheet based fluorescence correlation spectroscopy. | Opt Express. 2013 Apr 8;21(7):8652-68. |
| Ahrens MB, Keller PJ. | Whole-brain functional imaging at cellular resolution using light-sheet microscopy. | Nat Methods. 2013 Mar 18. doi: 10.1038/nmeth.2434. |
| Long F, Zeng S, Huang ZL. | Localization-based super-resolution microscopy with an sCMOS camera part II: experimentalmethodology for comparing sCMOS with EMCCD cameras. | Opt Express. 2012 Jul 30;20(16):17741-59. |
| Baumgart E, Kubitscheck U. | Scanned light sheet microscopy with confocal slit detection. | Opt Express. 2012 Sep 10;20(19):21805-14. |
| Takao D, Nemoto T, Abe T, Kiyonari H, Kajiura-Kobayashi H, Shiratori H, Nonaka S. | Asymmetric distribution of dynamic calcium signals in the node of mouse embryo during left-right axis formation. | Dev Biol. 2013 Apr 1;376(1):23-30. |
| Huang F, Hartwich TM, Rivera-Molina FE, Lin Y, Duim WC, Long JJ, Uchil PD, Myers JR, Baird MA, Mothes W, Davidson MW, Toomre D, Bewersdorf J. | Video-rate nanoscopy using sCMOS camera-specific single-molecule localization algorithms. | Nat Methods. 2013 May 26. doi: 10.1038/nmeth.2488. |
| Ma H, Kawai H, Toda E, Zeng, S, Huang ZL | Localization-based super-resolution microscopy with an sCMOS camera part III: camera embedded data processing significantly reduces the challenges of massive data handling | Optics Letters, Vol. 38, Issue 11, pp. 1769-1771 (2013) |
| Baumgart E., Kaminski T. | Scanned LSFM with Confocal Detection. | Imaging and Microscopy, Volume 15, Issue 1 2013. |
| Xu D, Jiang T, Li A, Hu B, Feng Z, Gong H, Zeng S, Luo Q. | Fast optical sectioning obtained by structured illumination microscopy using a digital mirror device. | J Biomed Opt. 2013 Jun;18(6):60503. doi: 10.1117/1.JBO.18.6.060503. |
PC recommendations
With the introduction of the ORCA-Flash4.0 V3, users are now able to stream 4 megapixel images to their computers 100 frames per second. The computer recommendations for this high data rate can be met by using the guidelines listed in the PC Recommendations for ORCA-Flash4.0 V3.
Software
Our software provides the interface to access all our carefully engineered camera features, from simply setting exposure to orchestrating complex triggering for multidimensional experiments.
Specifications
| Type number | C13440-20CU |
|---|---|
| Imaging device | sCMOS |
| Effective no. of pixels | 2048 (H)×2048 (V) |
| Cell size | 6.5 μm×6.5 μm |
| Effective area | 13.312 mm×13.312 mm |
| Full well capacity | 30 000 electrons (typ.) * |
| Readout speed | 100 frames/s (Full resolution, standard scan, Camera Link) * 40 frames/s (Full resolution, standard scan, USB 3.0, 16 bit) * 53 frames/s (Full resolution, standard scan, USB 3.0, 12 bit) * 80 frames/s (Full resolution, standard scan, USB 3.0, 8 bit) * |
| Readout noise | Standard scan (at 100 frames/s, typ.):1.6 electrons rms (1.0 electrons median) Slow scan (at 30 frames/s, typ.): 1.4 electrons rms (0.8 electrons median) |
| Cooling method | Peltier cooling |
| Cooling temperature | Forced air (Ambient at +20 ℃): -10 ℃ Water (+20 ℃): -10 ℃ Water (+15 ℃): -30 ℃ |
| Dark current | 0.06 electrons/pixel/s (Air Cooled to -10° C) (typ.) 0.06 electrons/pixel/s (Water Cooled to -10° C) (typ.) 0.006 electrons/pixel/s (Water Cooled to -30° C) (typ.) |
| Dynamic range | 37 000:1 (typ.) |
| Interface | Camera Link / USB 3.0 |
| A/D converter | 16 bit / 12 bit / 8 bit |
| Lens mount | C-mount |
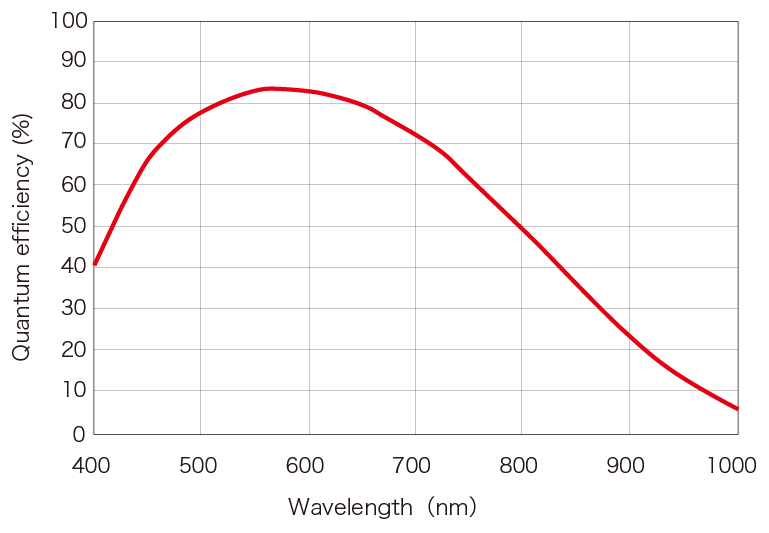
Spectral response
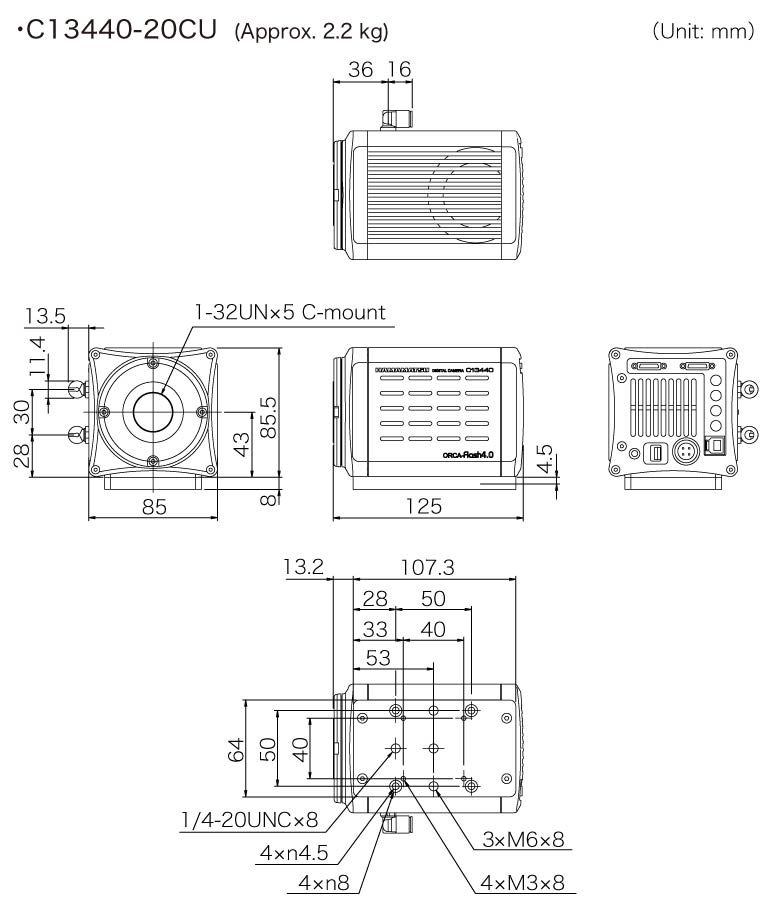
Dimensions
Instruction manual
Related documents
- Confirmation
-
It looks like you're in the . If this is not your location, please select the correct region or country below.
You're headed to Hamamatsu Photonics website for US (English). If you want to view an other country's site, the optimized information will be provided by selecting options below.
In order to use this website comfortably, we use cookies. For cookie details please see our cookie policy.
- Cookie Policy
-
This website or its third-party tools use cookies, which are necessary to its functioning and required to achieve the purposes illustrated in this cookie policy. By closing the cookie warning banner, scrolling the page, clicking a link or continuing to browse otherwise, you agree to the use of cookies.
Hamamatsu uses cookies in order to enhance your experience on our website and ensure that our website functions.
You can visit this page at any time to learn more about cookies, get the most up to date information on how we use cookies and manage your cookie settings. We will not use cookies for any purpose other than the ones stated, but please note that we reserve the right to update our cookies.
1. What are cookies?
For modern websites to work according to visitor’s expectations, they need to collect certain basic information about visitors. To do this, a site will create small text files which are placed on visitor’s devices (computer or mobile) - these files are known as cookies when you access a website. Cookies are used in order to make websites function and work efficiently. Cookies are uniquely assigned to each visitor and can only be read by a web server in the domain that issued the cookie to the visitor. Cookies cannot be used to run programs or deliver viruses to a visitor’s device.
Cookies do various jobs which make the visitor’s experience of the internet much smoother and more interactive. For instance, cookies are used to remember the visitor’s preferences on sites they visit often, to remember language preference and to help navigate between pages more efficiently. Much, though not all, of the data collected is anonymous, though some of it is designed to detect browsing patterns and approximate geographical location to improve the visitor experience.
Certain type of cookies may require the data subject’s consent before storing them on the computer.
2. What are the different types of cookies?
This website uses two types of cookies:
- First party cookies. For our website, the first party cookies are controlled and maintained by Hamamatsu. No other parties have access to these cookies.
- Third party cookies. These cookies are implemented by organizations outside Hamamatsu. We do not have access to the data in these cookies, but we use these cookies to improve the overall website experience.
3. How do we use cookies?
This website uses cookies for following purposes:
- Certain cookies are necessary for our website to function. These are strictly necessary cookies and are required to enable website access, support navigation or provide relevant content. These cookies direct you to the correct region or country, and support security and ecommerce. Strictly necessary cookies also enforce your privacy preferences. Without these strictly necessary cookies, much of our website will not function.
- Analytics cookies are used to track website usage. This data enables us to improve our website usability, performance and website administration. In our analytics cookies, we do not store any personal identifying information.
- Functionality cookies. These are used to recognize you when you return to our website. This enables us to personalize our content for you, greet you by name and remember your preferences (for example, your choice of language or region).
- These cookies record your visit to our website, the pages you have visited and the links you have followed. We will use this information to make our website and the advertising displayed on it more relevant to your interests. We may also share this information with third parties for this purpose.
Cookies help us help you. Through the use of cookies, we learn what is important to our visitors and we develop and enhance website content and functionality to support your experience. Much of our website can be accessed if cookies are disabled, however certain website functions may not work. And, we believe your current and future visits will be enhanced if cookies are enabled.
4. Which cookies do we use?
There are two ways to manage cookie preferences.
- You can set your cookie preferences on your device or in your browser.
- You can set your cookie preferences at the website level.
If you don’t want to receive cookies, you can modify your browser so that it notifies you when cookies are sent to it or you can refuse cookies altogether. You can also delete cookies that have already been set.
If you wish to restrict or block web browser cookies which are set on your device then you can do this through your browser settings; the Help function within your browser should tell you how. Alternatively, you may wish to visit www.aboutcookies.org, which contains comprehensive information on how to do this on a wide variety of desktop browsers.
5. What are Internet tags and how do we use them with cookies?
Occasionally, we may use internet tags (also known as action tags, single-pixel GIFs, clear GIFs, invisible GIFs and 1-by-1 GIFs) at this site and may deploy these tags/cookies through a third-party advertising partner or a web analytical service partner which may be located and store the respective information (including your IP-address) in a foreign country. These tags/cookies are placed on both online advertisements that bring users to this site and on different pages of this site. We use this technology to measure the visitors' responses to our sites and the effectiveness of our advertising campaigns (including how many times a page is opened and which information is consulted) as well as to evaluate your use of this website. The third-party partner or the web analytical service partner may be able to collect data about visitors to our and other sites because of these internet tags/cookies, may compose reports regarding the website’s activity for us and may provide further services which are related to the use of the website and the internet. They may provide such information to other parties if there is a legal requirement that they do so, or if they hire the other parties to process information on their behalf.
If you would like more information about web tags and cookies associated with on-line advertising or to opt-out of third-party collection of this information, please visit the Network Advertising Initiative website http://www.networkadvertising.org.
6. Analytics and Advertisement Cookies
We use third-party cookies (such as Google Analytics) to track visitors on our website, to get reports about how visitors use the website and to inform, optimize and serve ads based on someone's past visits to our website.
You may opt-out of Google Analytics cookies by the websites provided by Google:
https://tools.google.com/dlpage/gaoptout?hl=en
As provided in this Privacy Policy (Article 5), you can learn more about opt-out cookies by the website provided by Network Advertising Initiative:
http://www.networkadvertising.org
We inform you that in such case you will not be able to wholly use all functions of our website.
Close